|
The sample may either transmit or absorb each different wavelength of this incoming light transmitted light passes through the sample, while absorbed light gets taken in by the sample. They place this sample into the spectrophotometer, which then floods it with a continuous stream of light particles. Say, for example, scientists want to determine the components of a reaction product. Let’s go over this process and its implications in more detail. When this light contacts the substance, it separates into its various wavelengths Scientists place cuvettes of different materials into the machine, which passes light through the samples. The spectrophotometer essentially functions as a glorified prism. 
When visible light passes through a prism, its component wavelengths split due to their varying energies and travelling speeds. Exploring the SpectrophotometerĬolors and associated energies are therefore two key components of spectrophotometric investigations. For example, as measured in nanometers (nm), blue and purple light exhibit shorter wavelengths, while red light displays longer wavelengths. Shorter wavelengths denote higher energy levels, while longer wavelengths correspond to lower energy levels. The color of traveling light corresponds to the lengths of its photon-comprised waves, which relate to the energies of these photons. This flow of photons resembles the shape of a wave because this electromagnetic wave is composed of discrete parts, or photons, it is not continuous. These photons move so fast that, in a single second, each covers 180,000 miles. Sources of light emit particles called photons. Below, we cover the mechanisms and applications of spectrophotometry in more detail. 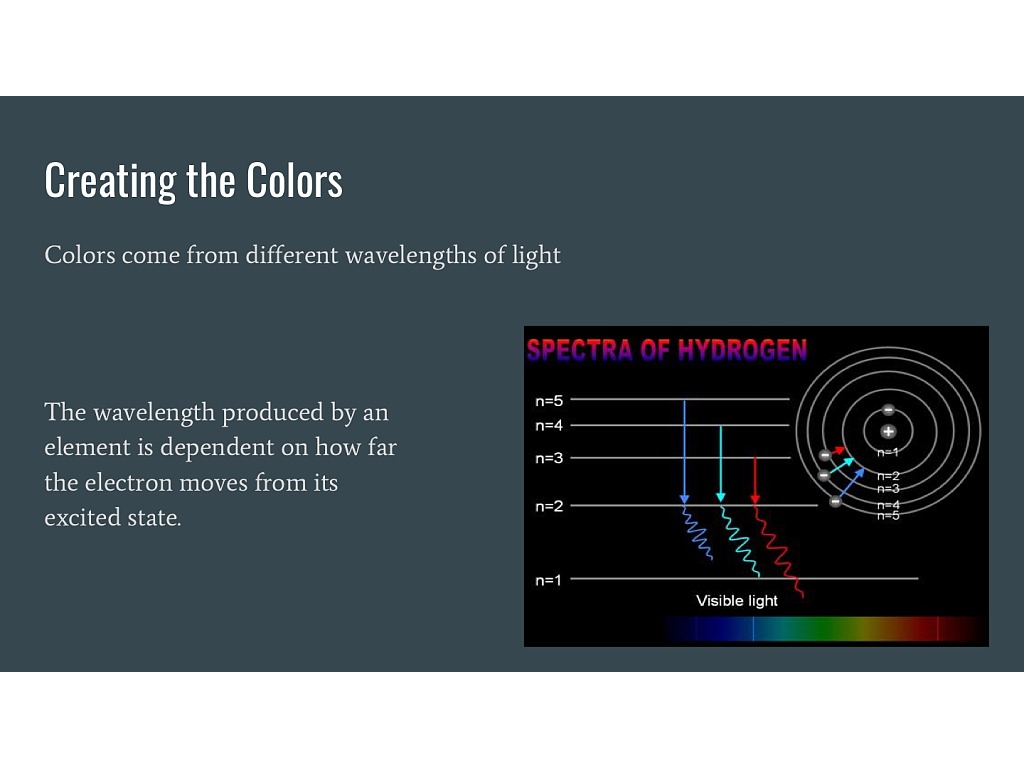
This process provides insight into protein synthesis, DNA and RNA function, bacterial cell growth, enzymatic reactions, and more. Spectrophotometry involves the measurement of a substance’s concentration, amount, and identity. It facilitates developments in energy, pharmaceutical, health care, agricultural, manufacturing, and forensic realms. Spectrophotometry informs research in the physical sciences, including chemistry, biology, physics, geology, and astronomy. Introduction to Spectrophotometry and Emission Spectrum Spectrophotometer: machine that quantifies the intensity of light transmitted and emitted by a substance, indirectly informing the identification and measurement of the substance.Prism: transparent, triangular device that bends light, revealing its different wavelengths and their corresponding energies.Cuvette: a spectrophotometry-specific piece of glassware with straight, clear sides holds samples during spectrophotometric analyses.Photon: particle containing a distinct quantum, or amount, of light energy basic unit of light.Discovering the Electron: JJ Thomson and the Cathode Ray Tube.You will also learn about spectrophotometry and the mechanisms involved, as well as its applications to scientific research. You will also learn how emission spectra is produced. To understand that the spectral emission lines of an element have the same frequencies as the lines of the absorption spectrum.In this tutorial, you will learn about emission spectrum, absorption spectrum, and what is a spectrophotometer.To characterize an atom by its light spectrum and to introduce spectroscopy.To distinguish between absorption spectra and emission spectra.CRC Handbook of Chemistry and Physics NSRDS-NBS 68 (1980).Ĭlick on an element to select it. 
Source for the values of spectral lines: CDS Strasbourg University ( link) from Reader J., and Corliss Ch.H. For this reason, a gas composed of a single atom can absorb or emit a limited number of frequencies.įor a given element, the emission spectrum (upper part of the animation) has the same frequency as its absorption spectrum (bottom part). It is "quantized" (see animation line spectrum of the hydrogen atom). One of the great discoveries of quantum mechanics is that the energy of an atom can only have certain well-defined values. The spectrum obtained can be continuous or discrete ("line spectrum"). A prism (or an array) is used to break a beam of light according to its different frequencies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
